 
The answer that this is because half-filled subshells are intrinsically more stable, although given often, is not correct- see this paper (or this one) for an in-depth view. 
Why does this happen? I asked my teacher and she could not give an answer, but she guessed it had something to do with the stability of the electron orbitals.Īh, I remember this question from when I TA'd General Chemistry.  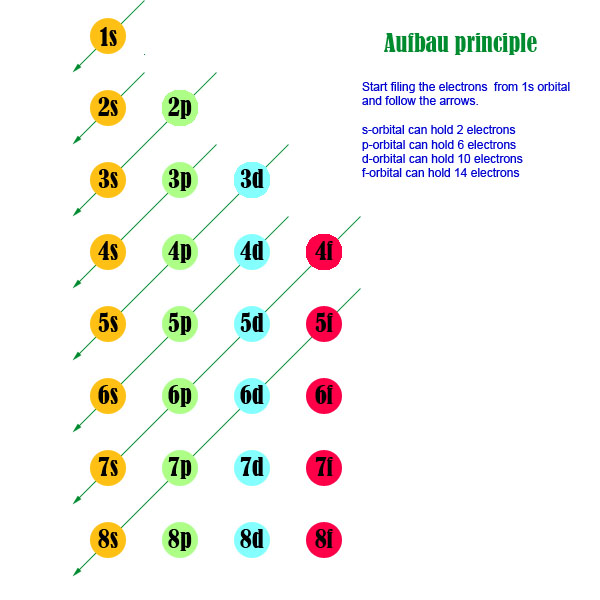
This anomaly also appears to occur in copper. It in fact has an electron arrangement of 2,8,13,1 and an electronic configuration of 1s 2 ,2s 2, 2p 6, 3s 2 ,3p 6 ,4s 1 ,3d 5 -even though this seems to defy the Aufbau principle. Vanadium has the electron arrangement 2,8,11,2 and the electronic configuration 1s 2 ,2s 2, 2p 6, 3s 2 ,3p 6 ,4s 2 ,3d 3 - so by the Aufbau principle you would expect Chromium, the next element, to have an electron arrangement of 2,8,12,2 and an electron configuration of 1s 2 ,2s 2, 2p 6, 3s 2 ,3p 6 ,4s 2 ,3d 4 (since 4s fills before 3d), but it does not. We were writing out the electronic configuration of the first 30 elements and I noticed something weird when I came to Chromium. Hello guys! I have a question about the filling of electron shells as you go along the period of the periodic table. We make our world significant by the courage of our questions and by the depth of our answers. For more open-ended questions, try /r/AskScienceDiscussion | Sign up to be a panelist!.Looking for flair? Sign up to be a panelist!.Neuroscience, Neurology, Neurochemistry, Cognitive NeuroscienceĪskScience AMA Series: NASA Software CatalogĪsk Anything Wednesday - Physics, Astronomy, Earth and Planetary ScienceĪskScience AMA Series: Extreme Heat, PNNLĪsk Anything Wednesday - Engineering, Mathematics, Computer scienceĪsk Anything Wednesday - Biology, Chemistry, Neuroscience, Medicine, PsychologyĪsk Anything Wednesday - Economics, Political Science, Linguistics, AnthropologyĪskScience AMA Series: Squidtember, OceanX Medicine, Oncology, Dentistry, Physiology, Epidemiology, Infectious Disease, Pharmacy, Human Body Psychology, Cognitive Psychology, Developmental Psychology, Abnormal, Social Psychology Social Science, Political Science, Economics, Archaeology, Anthropology, Linguisticsīiology, Evolution, Morphology, Ecology, Synthetic Biology, Microbiology, Cellular Biology, Molecular Biology, Paleontology Mechanical Engineering, Electrical Engineering, Structural Engineering, Computer Engineering, Aerospace EngineeringĬhemistry, Organic Chemistry, Polymers, Biochemistry Mathematics, Statistics, Number Theory, Calculus, AlgebraĪstronomy, Astrophysics, Cosmology, Planetary FormationĬomputing, Artificial Intelligence, Machine Learning, ComputabilityĮarth Science, Atmospheric Science, Oceanography, Geology Theoretical Physics, Experimental Physics, High-energy Physics, Solid-State Physics, Fluid Dynamics, Relativity, Quantum Physics, Plasma Physics /r/AskScienceDiscussion: For open-ended and hypothetical questions.FAQ: In-depth answers to many popular questions.Weekly Features: Archives of AskAnything Wednesday, FAQ Fridays, and more!.Be civil: Remember the human and follow Reddiquette.Report comments that do not meet our guidelines, including medical advice.Downvote anecdotes, speculation, and jokes.Upvote on-topic answers supported by reputable sources and scientific research.Answer questions with accurate, in-depth explanations, including peer-reviewed sources where possible.Please read our guidelines and FAQ before posting 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
